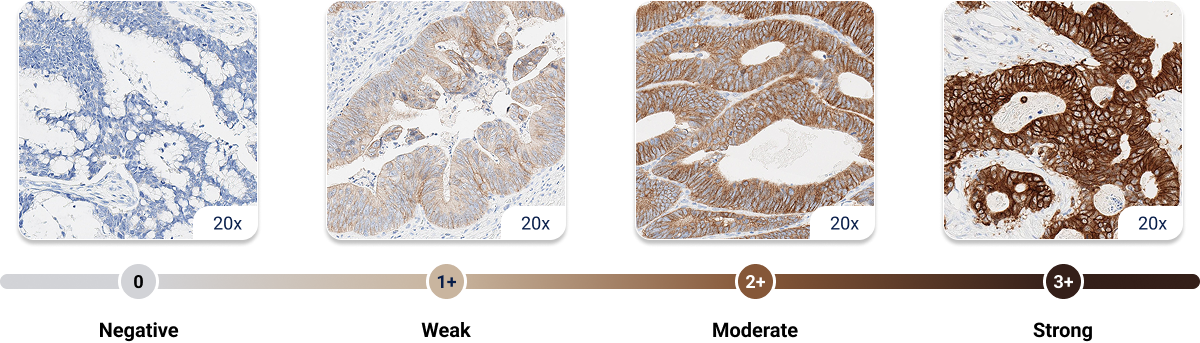
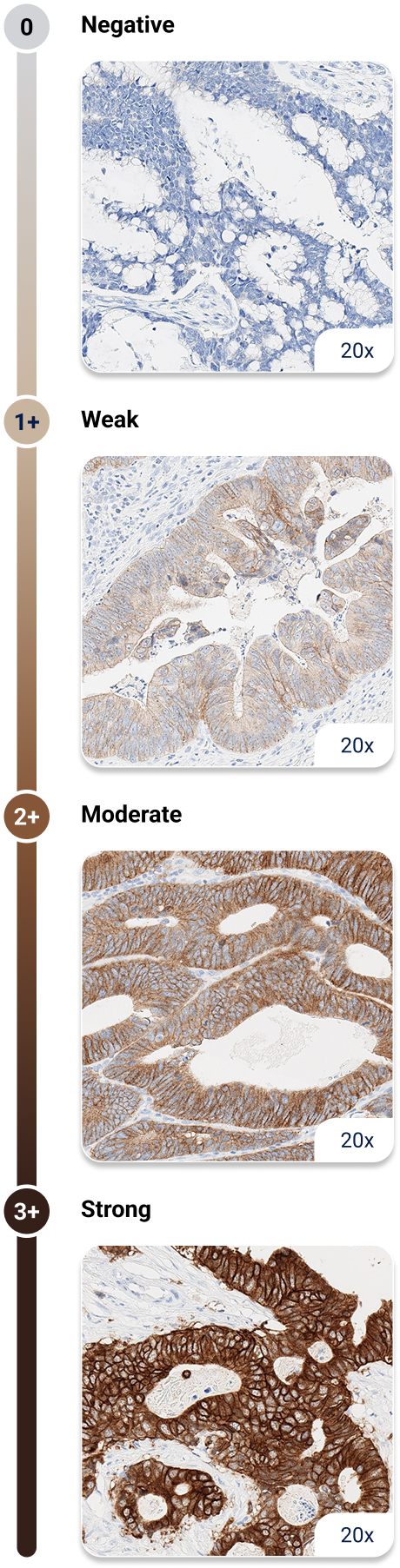
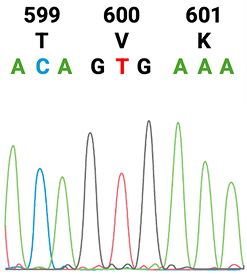
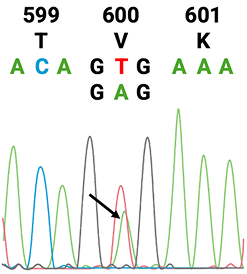
1. American Cancer Society. Colorectal Cancer Early Detection, Diagnosis, and Staging. January 29, 2024. Accessed August 15, 2025.
https://www.cancer.org/cancer/types/colon-rectal-cancer/detection-diagnosis-staging/staged.html.
2. Wang R, et al.
World Gastrointest Surg. 2023;15(12):2907-2918.
doi:10.4240/wjgs.v15.i12.2907.
3. Adekunle AD, et al.
JAMA Netw Open. 2024;7(7):e2419110.
doi:10.1001/jamanetworkopen.2024.19110.
4. Kasi A, et al.
Curr Colorectal Cancer Rep. 2020;16(5):97-106.
doi:10.1007/s11888-020-00458-z.
5. Saridaki Z, et al.
Br J Cancer. 2015;113(6):914-920.
doi:10.1038/bjc.2015.307.
6. Armstrong GR, et al.
Int J Exp Pathol. 2021;102(3):172-178.
doi:10.1111/iep.12395.
7. Suwaidan AA, et al.
Cancer Treat Rev. 2022;105:102363.
doi:10.1016/j.ctrv.2022.102363.
8. Lee MKC, Loree JM.
Curr Oncol. 2019;26(Suppl 1):S7-S15.
doi:10.3747/co.26.5719.
9. Jan YH, et al.
Br J Cancer. 2022;127(7):1304-1311.
doi:10.1038/s41416-022-01913-4.
10. Pietrantonio F, et al.
Ann Oncol. 2018;29(6):1394-1401.
doi:10.1093/annonc/mdy090.
11. Qamsari ES, et al.
Tumor Biol. 2017;39(5):101042317699118.
doi:10.1177/101042831769911.
12. Alese OB, et al.
Am Soc Clin Oncol Educ Book. 2023;43:e389574.
doi:10.1200/EDBK_389574.
13. Hawes D, et al.
Mod Surg Pathol. 2009:48-70.
doi:10.1016/B978-1-4160-3966-2.00016-3.
14. McCampbell AS, et al.
Appl Immunohistochem Mol Morphol. 2019;27(5):345-355.
doi:10.1097/PAI.0000000000000593.
15. Fedchenko N, Reifenrath J.
Diagn Pathol. 2014;9:221.
doi:10.1186/s13000-014-0221-9.
16. Kim SW, et al.
J Pathol Transl Med. 2016;50(6):411-418.
doi:10.4132/jptm.2016.08.08.
17. Bradley CA, et al.
Oncotarget. 2016;7(48):78932-78945.
doi:10.18632/oncotarget.12933.
18. Lee SJ, et al.
Clin Colorectal Cancer. 2018;17(3):165-169.
doi:10.1016/j.clcc.2018.02.013.
19. Ha SY, et al.
Mod Pathol. 2013;26(12):1632-1641.
doi:10.1038/modpathol.2013.108.
20. Lin CL, et al.
Life (Basel). 2021;11(9):890.
doi:10.3390/life11090890.
21. Vendrell JA, et al.
Oncotarget. 2017;8(25):40345-40358.
doi:10.18632/oncotarget.15875.
22. NGS Workflow and Fundamentals of Sample Preparation. Enzo. Accessed December 1, 2025.
https://www.enzo.com/note/ngs-workflow-and-fundamentals-of-sample-preparation/.
23. Ihle MA, et al.
BMC Cancer. 2014;14:13.
doi:10.1186/1471-2407-14-13.
24. Sarhadi VK, Armengol G.
Biomolecules. 2022;12(8):1021.
doi:10.3390/biom12081021.
25. Ruiz C, et al.
Hum Mutat. 2020;41(5):1051-1068.
doi:10.1002/humu.23987.
26. Khehra N, et al. National Library of Medicine, National Center for Biotechnology Information. Polymerase Chain Reaction (PCR). March 6, 2023. Accessed July 31, 2025.
https://www.ncbi.nlm.nih.gov/books/NBK589663/.
27. From RNA to results: the essential guide to RT-PCR. Integra Biosciences. Accessed December 1, 2025.
https://www.integra-biosciences.com/global/en/blog/article/rna-results-essential-guide-rt-pcr.
28. Real-time PCR-based BRAF mutation detection. TrimGen Genetic Diagnostics. Accessed December 1, 2025.
https://www.trimgen.com/products/braf-mutation-detection-reagent-kit-real-time-PCR.